Are there trade implications if the UK starts growing gene-edited crops and the EU doesn't? Could gene-edited crops be grown alongside conventional and organic crops? Why does the NFU support gene editing? How do opinions differ between the devolved nations?
The Genetic Technology (Precision Breeding) Bill has been passed into law after its passage passage through Parliament and following a three-month public consultation in 2021.
NFU Vice President David Exwood said: “For more than two decades, we’ve advocated for the development of biotechnology within agriculture and horticulture, and for science to dictate the policy."
I am delighted that, after working closely with Defra, the Food Standards Agency, parliamentarians, and industry partners to ensure the Bill’s smooth progress, it has passed its final hurdle.
NFU signs joint welfare statement
The NFU was one of the signatories on an open statement of support for the Genetic Technology (Precision Breeding) Bill.
Organisations and individuals from across the scientific, breeding, farming, veterinary and input supply sectors have come together to express support for the potential role of new breeding technologies such as gene editing in delivering more sustainable, high-welfare UK livestock research and production.
The statement was coordinated by the All-Party Parliamentary Group on Science and Technology in Agriculture ahead of the Bill's second reading on 21 November 2022.
What is the difference between GM (gene modification) and GE (gene editing)?
GM technologies involve inserting new DNA into an organism’s genome, giving the resulting plant or animal desired characteristics. Gene editing is a group of technologies that make the changes within the organism’s own DNA by moving, adding or deleting precise pieces of genetic material. Such changes are what happens randomly in conventional breeding, and can also come from mutations induced by chemicals or gamma rays, as with pink grapefruit or the malting barley variety, Golden Promise.
Watch wheat geneticist Cristobal Uauy explaining crop breeding and gene editing to the 2019 Oxford Farming Conference. This video from UC Davies explains livestock gene editing in two minutes. You’ll find plenty more talks like these on YouTube.
How is gene editing currently used in agriculture and horticulture?
Two examples of crops currently authorised for commercial distribution that use GE technology and are not regulated as GMOs are:
- Calyno, a healthier oil (high oleic, low linolenic resulting in reduced trans-fatty acids associated with cardiovascular disease) which is produced from a variety of gene-edited soybean and has been on the US market since 2019.
- In December 2020, Japan authorised a genome-edited tomato with higher levels of a blood pressure-lowering compound. The Sicilian Rouge High GABA tomato is now on sale and is voluntarily labelled as GE.
Products in the next few years from technology company Calyxt are likely to include a high-fibre wheat and a reduced-browning potato. UK based Tropic Biosciences are also working on development of a non-browning banana.
In the USA, Conscious Greens, a milder mustard green for use in salads, are currently going through consumer testing with the aim to land in supermarkets later this year.
What possible applications are there for gene editing in farming?
There are gene-editing applications in at least 46 different crop species, with rice, tobacco, tomato, maize, wheat and soybean among the most cited. A very broad range of products with market-oriented traits are being developed, and not only those with agronomic traits - such as yield and disease resistance - but also foods with consumer-facing traits, such as lower allergenicity, reduced contaminants, high anti-oxidant, longer shelf life, vitamin enhanced, heart-healthy. There are also those with climate-resilience traits, such as drought and salt tolerance.
Most recently, scientists at Rothamsted Research and the University of Bristol have used gene editing to develop a type of wheat that is less likely to produce carcinogenic acrylamide when baked. The government has approved a series of field trials of the CRISPR-edited wheat, which has been bred to have lower levels of the amino acid asparagine. The first trial started in autumn 2021, the first such trial of gene-edited wheat to be carried out anywhere in Europe.
In livestock, gene-editing applications being developed in research include: porcine reproductive and respiratory syndrome virus (PRRSV) and African swine flu resistance in pigs; bTB resistance in cattle; mastitis resistance; hornless cattle; chickens that cannot spread bird flu; elimination of milk allergen; increased lean muscle.
Why does the NFU support gene editing?
The central principle of NFU biotechnology policy is choice to access the best available tools to farm sustainably and profitably. Biotechnology is not a silver bullet, but the NFU sees great potential in new precision breeding techniques, such as gene editing, to tackle challenges associated with climate change, nutrition and crop and livestock disease. We would therefore like to see more R&D in applications of value to British food and farming; and a well-functioning, fit-for-purpose, evidence-based legislative process to regulate their use.
Much more detail on, and the evidence behind the NFU’s policy position, can be found in our response to the 2021 Defra consultation.

How are GMO (genetically modified organisms) and gene editing currently regulated?
To protect human health, the environment and to ensure consumer choice, there is strict legislation controlling the deliberate release into the environment of GMOs.
There are three main parts to legislation of GMOs at EU level:
- Deliberate release
- Food and feed
- Traceability and labelling
Following Brexit, the UK retained these in domestic law through the Environmental Protection Act, and separate regulations on deliberate release, food, animal feed and traceability and labelling.
It is a devolved area of law and in March 2021 some initial changes were made to the law for England to facilitate R&D in plants that are not GMOs.
The FSA has responsibility for risk analysis in the authorisation of GM food and feed with advice from its Advisory Committee on Novel Foods and Processes (and FSS for Scotland), with Defra and devolved administrations as the competent authorities.
Release of a GM organism or import and marketing of a GM product (for example, seed, food, feed, animal, vaccine) requires formal authorisation. The assessment process considers potential safety factors, such as toxicity, allergenicity, and any possible transfer of novel genes to other organisms. Applicants provide a dossier of relevant information and this is scrutinised by Defra’s independent ACRE (Advisory Committee on Releases to the Environment).
Legislators here, in the EU and in many countries around the world are now actively reviewing how new breeding techniques such as gene editing should be regulated. For the EU, in July 2018, the European Court ruled that any and all mutagenesis breeding technologies should be regulated as a GMO, except those with long history of safe use (but MS could regulate these as GM too if they wish).
At the time, the UK strongly opposed the ruling, as did many organisations and countries within the EU. At the end of 2019, the EU Council asked the EU Commission to review the impact of the ruling, to analyse the link between biotechnology and the Green Deal Farm to Fork Strategy, and recommend how GE should be regulated. The study was published at the end of April 2021, with an initial focus on plants. Consultations, impact assessments and further policy action are progressing quite rapidly.
A public consultation on NGTs (New Genomic Techniques) in spring 2022 led to the EU Commission tabling a proposal (Plants produced by new genomic techniques) in July 2023 for a regulation on certain NGTs. It establishes two categories of plants obtained by NGTs: those comparable to naturally occurring or conventional plants, and those with modifications that are more complex. The two categories will be subject to different requirements to reach the market, taking into account their different characteristics and risk profiles.
The UK government responded to the Defra consultation in September 2021, setting out a step-by-step approach starting with improving the rules for research in GE plants and moving on to reviewing the regulatory definitions of a GMO to exclude certain organisms produced by gene editing and other genetic technologies if they could have been developed by traditional breeding.
On 25 May 2022, a Genetic Technology (Precision Breeding) Bill was introduced to Parliament “to ensure that plants, animals, and food and feed products developed using precision breeding technologies are regulated proportionately to risk. It will introduce simpler regulatory measures to enable these products to be authorised and brought to market more easily.” (Source: Defra fact sheet May 2022). This became the Genetic Technology (Precision Breeding) Act in March 2023, and work is now underway to set the legislative framework and processes to implement the Act, expected to be in place by the end of 2024 for plants and in 2025 for animals.
Are there differences of opinion between the devolved nations of the UK when it comes to gene editing?
The NFU is aware that regulation in this area is a devolved matter. However, the UK internal movement of goods is determined by the UK Internal Market (UKIM) Act, passed in 2020. In practice, this means that goods produced using methods legal in one part of the UK but not in another can still freely move throughout the UK (but noting that the Northern Ireland Protocol overrides the UKIM).
Furthermore, goods legally imported into one part of the UK can then freely move throughout then UK. In terms of gene editing, this means that should England authorise the use of gene editing in agricultural production, but Scotland and Wales do not, then goods produced in England using gene editing techniques can legally be sold in the other nations. This may lead to a competitive disadvantage for Scottish and Welsh businesses.
We also note the Scottish government and Welsh Assembly government have both been clear in their opposition to cultivation of GM crops in their countries. Imminent elections, Defra’s action plan post-consultation and what happens in the EU will all impact whether or not devolved administrations differentiate between GM and GE.
ScotGov originally said it will wait for the outcome of the EU Commission study on future regulation of GE and is expected to align with the EU position, while the SNP is reported as having softened its stance on GE.
In Wales, Minister of Environment, Energy and Rural Affairs Lesley Griffiths has also stated that Wales will maintain a precautionary approach to genetic modification, including gene editing.
The Northern Ireland position on gene editing is not entirely clear, but in 2015 Stormont decided to not allow GM cultivation, when EU member states were given sovereignty to do so; and the chairman of Stormont’s Committee on Agriculture, Environment and Rural Affairs was cautious in his reaction to the consultation, citing concerns about divergence and disruption. As the Genetic Technology (Precision Breeding) Bill was introduced to Parliament in May 2022, Defra Secretary of State George Eustice wrote to Scottish and Welsh administrations urging them to embrace the opportunities and join together to bring forward the legislation.
Why are the changes in regulation government is developing needed?
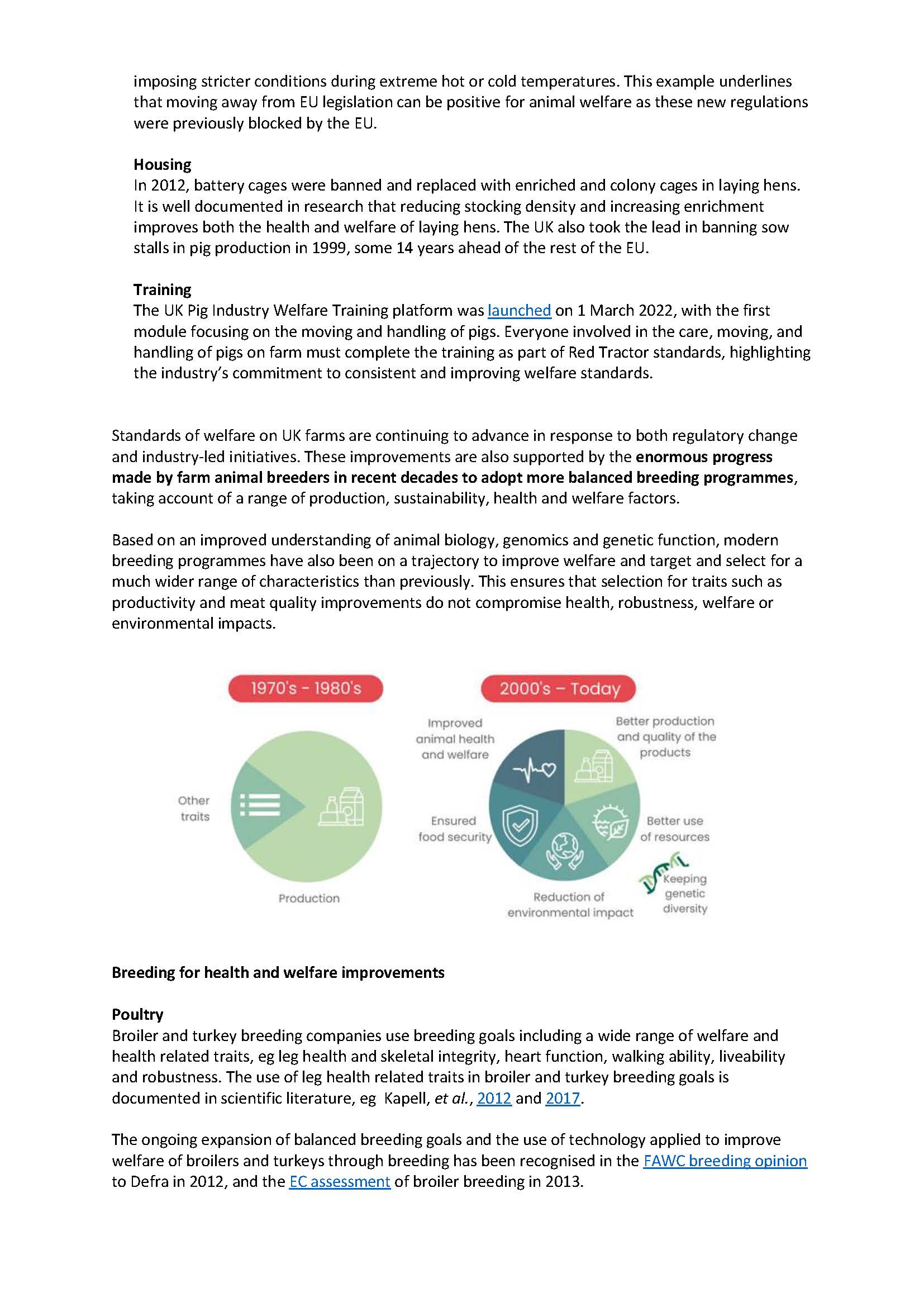
The science of genetic improvement is fast moving and new breeding techniques, such as gene-editing, are now widely used in the research community and are emerging commercially around the world.
The way these NBTs should be regulated is the subject of debate and regulatory reform globally driven by the fact that the majority cannot be described as GM as currently defined. There is a recognition of the particular potential value of these techniques to tackle intractable challenges and the important role of legislative frameworks in enabling R&D and access. Experience from Argentina shows that a separate approach for GE has led to a greater diversity of crops and traits and greater access by SMEs and public sector research.
In the EU, the ECJ ruled in July 2018 that any and all mutagenesis breeding technologies should be regulated as a GMO, except those with long history of safe use. There was consternation across EU institutions, member states, industry and the science community at the negative impact on innovation and sustainable agriculture. The EU Council asked the EU Commission to review the impact of the ruling, to analyse the link between biotechnology and the Green Deal Farm to Fork Strategy, and recommend how GE should be regulated. The study was published at the end of April 2021, with an initial focus on plants, and the EU Commission is working towards a new legislative proposal in 2023.
In November 2020, an EFSA assessment concluded that gene editing techniques do not pose more hazards than conventional or GM breeding. It concluded that existing risk assessments are applicable but with fewer data requirements.
In January 2021, Defra consulted on a proposal that, for England, it sets aside the 2018 European Court ruling and aligns with other countries such as Argentina, Australia, Brazil and Japan.
In September 2021, the government published its response, in light of the consultation responses it received. It states that it will take a step-by-step approach, starting by using existing powers under the Environmental Protection Act 1990 to facilitate R&D in plants, then bringing a Bill to parliament to provide a route to market, and finally reviewing the government’s approach to GMO regulation more broadly.
In March 2022, new legislation was approved that will make research and development easier for plants that have been produced by genetic technologies where the resulting genetic changes could have been developed using traditional breeding methods. Research scientists will continue to be required to notify Defra of these research trials. The commercial cultivation of these plants, and any food products derived from them, will still need to be authorised in accordance with existing GM rules.
In May 2022, a Genetic Technology (Precision Breeding) Bill was published “to ensure that plants, animals, and food and feed products developed using precision breeding technologies are regulated proportionately to risk. It will introduce simpler regulatory measures to enable these products to be authorised and brought to market more easily.” (Source: Defra fact sheet May 2022).
The four key policy changes are to:
- Remove plants and animals produced through precision breeding technologies from regulatory requirements applicable to the environmental release and marketing of GMOs (Genetically Modified Organisms).
- Introduce two notification systems; one for precision bred organisms used for research purposes and the other for marketing purposes. The information collected will be published on a public register on Gov.uk.
- Establish a proportionate regulatory system for the marketing of precision bred animals, to ensure animal welfare is safeguarded. We will not be introducing changes to the regulations for animals until this system is in place.
- Establish a new science-based authorisation process for food and feed products developed using precision bred organisms (FSA competency).
The Genetic Technology (Precision Breeding) Act passed into law in March 2023, and work is now underway to set the legislative framework and processes to implement the Act. It is expected this will be in place and open for applications to approve new PBOs by the end of 2024 for plants and in 2025 for animals.
The Food Standards Agency is responsible for regulating the use of PBOs in food and feed.
In Nov 2023 it published a consultation on this new framework [Consultation on proposals for a new framework in England for the regulation of precision bred organisms used for food and animal feed | Food Standards Agency]. The key elements are:
- A pre-market authorisation system designed around the classification of PBOs into two tiers requiring different levels of assessment, based on independent scientific advice relating to risk.
- A public register of PBOs for food/feed that have received marketing authorisation.
- Provisions for enforcement.
The NFU has submitted a response to the FSA’s consultation. Read our response in full.

Should gene-edited crops and livestock be regulated in the same way?
The underlying principles of fit-for-purpose and science-based regulation that ensure food, feed and environmental safety and animal welfare should be the same. The regulatory status of specific applications should be assessed on a case-by-case basis and in the context of existing legislation on breeding.
There are different rules and systems regulating breeding and production of crops and livestock, into which any new framework for gene editing would have to fit. Discussions are ongoing between government, industry and scientists about how to ensure proportionate protections and requirements are in place in terms of food and environmental safety, animal welfare, intellectual property and economic returns.
Are there any trade implications if the UK decides to align with international regulatory definitions of GM and gene editing while the EU maintains its current approach on GE?
The continued political nature of trade discussions both within UK and with EU makes it difficult to predict impact on market access and movement of goods. With robust risk management controls in place, ensuring health and environmental safety are not compromised, we believe the UK would continue to be able to trade with the EU on the basis of these rules. Other third countries, including major exporting nations such as Argentina, Brazil, Australia, Canada and the US, already regulate gene editing techniques differently to the EU. And for many years, the EU has relied heavily on GM protein feed from these countries.
There are reviews and consultations ongoing by governments around the world looking at how best to regulate gene editing and other new breeding techniques. In 2018, 13 countries issued a joint statement to the World Trade Organisation, stating that governments should “avoid arbitrary and unjustifiable distinctions” between those crops developed through precision breeding technologies, such as gene editing, and those developed through conventional breeding. Since then, the international regulatory environment for precision bred products has rapidly evolved and products derived through precision breeding have begun to enter the global market.
In practice, commercialisation of gene edited crops and livestock in the UK is many years away, by which time the EU will have set out a clearer and hopefully more pragmatic position based on the technological, commercial and regulatory situation around the world. Some EU-level organisations also responded to the Defra March 2021 consultation, with a link being made with how the EU will deliver its ‘Farm to Fork’ strategy and ‘Green Deal’ if new breeding technologies are strongly discouraged by inappropriate legislation. The EU Commission study published in April 2021 suggests such positive change will come, saying that current legislation is not fit for purpose for some new genetic technologies and their products, and that it must be adapted to keep pace with scientific and technological progress.
The legislative change is progressing well (Plants produced by new genomic techniques (europa.eu)), with proposals currently being discussed in Parliament led by the Committee on the Environment, Public Health and Food Safety (ENVI). The draft report is scheduled for presentation on 29 November 2023 and will be put to the vote on 24 January 2024.
Why do we need this in addition to supporting agroecology and integrated pest management (IPM)?
Breeding is an important part of both agroecological approaches and IPM, which are, by their nature, integrated and advocate using a range of different tools and practices. There are no silver bullets in farming. No one technology, system or practice provides all the solutions and the NFU supports R&D and enabling regulations to ensure options are open to all farmers now and in the future.
How could gene-edited crops be grown alongside conventional and organic crops?
If there end up being separate markets for crops and livestock based on the breeding technique used to produce them, this would be managed in the same way as it is currently. Coexistence, identity preservation and sales based on varieties or breeds are an established part of farming now, as well as the separate market for organic produce. The same principles and logistics would apply with gene edited products if the market demands it.
How will supply chains be able to identify and differentiate gene-edited varieties?
Transparency and openness are essential as these new breeding techniques are developed and used. However, there is no scientific basis for statutory segregation or labelling in terms of safety or detection for GE products where genetic changes could have occurred through conventional breeding. Indeed, there is a risk that consumers would be misled if GE was labelled on foods at point of sale.
Where such products are indistinguishable from their conventionally bred counterparts, we would question whether it is acceptable to expect consumers to pay additional costs of segregation when there is no evidence of additional risks.
Indeed, many of the gene edited traits and products under development are consumer health-focussed, such as gluten-free or low asparagine wheat; tomatoes with high vitamin D or blood-pressure lowering compounds; healthier vegetable oils; non-browning apple or potatoes. It is therefore likely that the resulting crop varieties will command a premium in the market place, such that segregation will be proactively maintained within the agri-food supply chain to protect that added value.
The latest FSA consultation on regulating PB food and feed points to the UK government position that there is no consumer safety justification for labelling on all PB food and no plans to require it by law. Labelling falls within the policy remit of Defra in England and, after much discussion during the passage of the Bill, there is no provision for labelling in the Act.
The Genetic Technology (Precision Breeding) Act will introduce a system such that any new precision-bred organism will be notified to government and information approved PBOs published on a public register. Any food or feed produced from a PBO must approved by FSA and will also be listed on a public register.
The British Society of Plant Breeders has committed to setting up a complementary public list of varieties produced using these breeding techniques. So, the information will be available for growers, the supply chain and consumers to make the choice and to label voluntarily if the market demands it.